II. The Chemistry of Life (Chapter 2: Background for Biochemistry)
A. Key Forces Acting on MatterElectromagnetic Force
The electromagnetic force accounts for the fact that positive particles repel and oppposites attract and it causes chemical reactions because when opposites attract and join therfore a chemical reation is caused. Nuclear Force The nuclear force causes the protons and electrons to not be able to touch and allows atoms to exist as we know them because the protons and electrons do not intefere or have any contact with each other. The nuclear force is greater force as evidenced by the ability to keep the outer electrons away atom the center of the atom. |
C. The Atom The Quantum Mechanical Model of the atom is the current theory of the atom that states that there is no boundary between particles and that it is impossible to know exactly how particles are shaped and how they move. In other words we will never be able to understand the atom completely, and it will always remain a mystery. Albert Einstein died trying to to discover all the unknown information about the atom.
|
B. MatterDefine in your own words, give examples, include images and source to
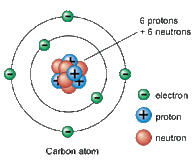
reference for basic chemistry terms needed to understand biochemistry 1.atoms: a basic unit of matter with a specific number of protons for each electron. Atoms have a neutral charge when isolated. This means that the number of protons will equal the number of electrons in an atom.
|
D. Sub-Atomic Particles
|
E. Chemical BondsDefine in your own words, give examples, include images and source to reference
for the following bond types (in order of increasing strength):
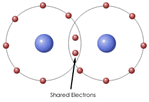
Covalent Bonds are formed when valence electrons in atomic
orbitals are shared. Examples of Covalently Bonded Molecules:
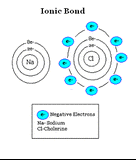
Examples of Ionically Bonded Molecules:
covalent bonds forms an electrostatic (charge, as in positive attracted to negative) interaction with a substance of opposite charge.
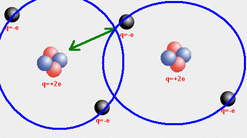
Van der Walls Forces are weak, short-range electrostatic attractive forces between uncharged molecules, arising from the interaction of permanent or transient electric dipole moments. Examples:
|
F. Water and It's Importance to LifeInclude the following in
your discussion about water:
|
Covalent Bonds:
Ionic Bond:
Metallic Bonds:
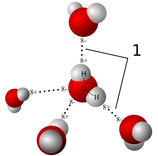
Hydrogen Bonds:
Van der Waals forces: